Helping regulatory teams navigate the structured data era
Practical tools and intelligence for Regulatory Information Managers and consultants on eCTD 4.0, IDMP, and the expanding landscape of Health Authority controlled vocabularies.
The Regulatory Data Challenge Facing Pharma
Global regulatory submissions are no longer driven only by documents. Increasingly, regulators require structured product data that must be consistent across multiple regulatory frameworks, including IDMP and eCTD 4.0.
For pharmaceutical companies, this creates a significant operational challenge. Product data, organisational roles, dose forms, strengths and other key attributes must be sourced from internal systems, managed through regulatory platforms, and aligned with regulator-specific controlled vocabularies.
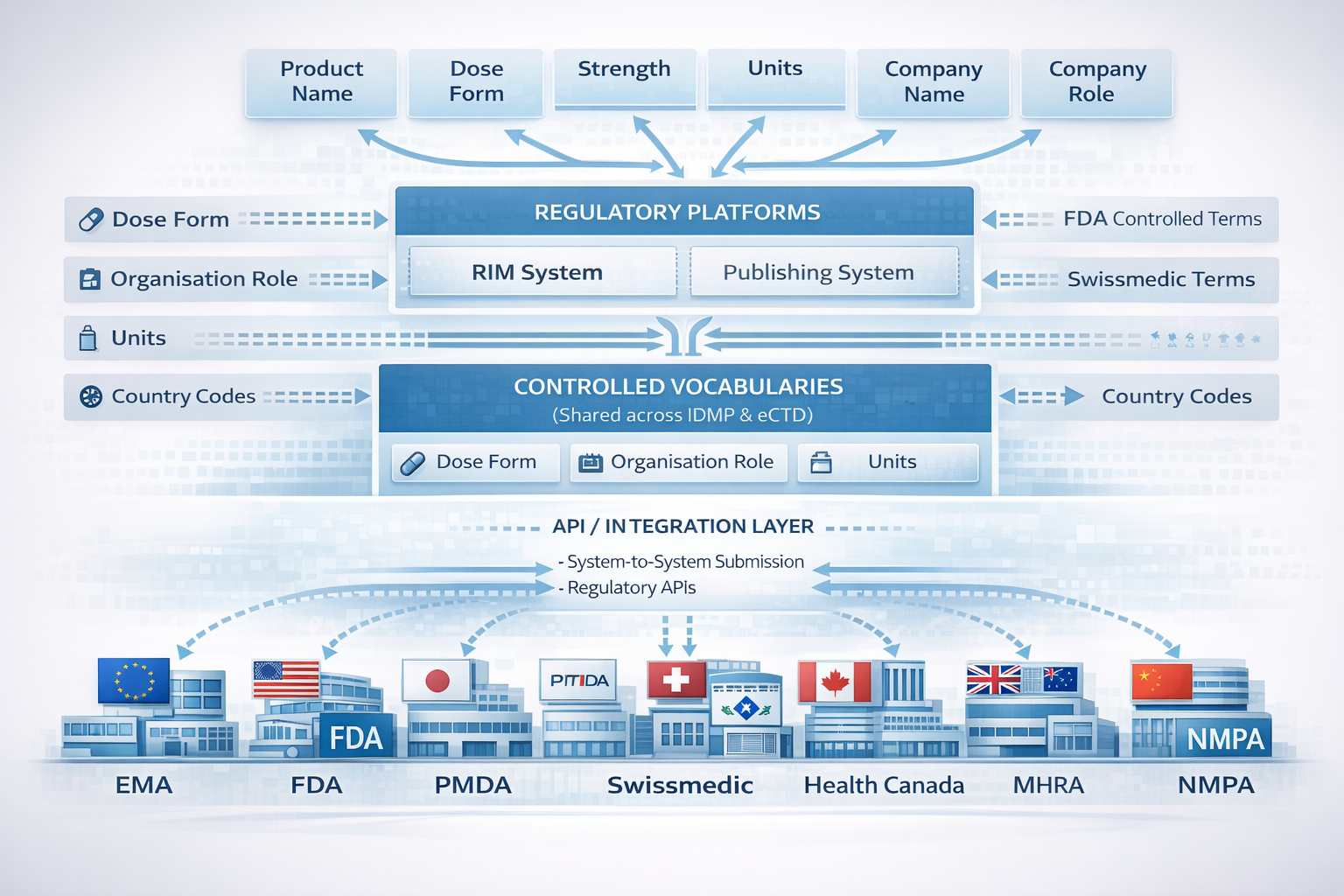
How regulatory data flows from internal business information through regulatory systems to global health authorities.
At the heart of this challenge is the need to manage core product and organisational data across multiple regulatory processes.
These systems transform internal business data into submission-ready information aligned to the regulatory frameworks used by global authorities. A further complication arises because regulators often require the use of controlled vocabularies, many of which are shared across IDMP and eCTD but implemented differently by each authority.
The result is a complex ecosystem of systems, standards and regulatory requirements that must work together seamlessly.
Core business data that must be governed centrally
- Product name
- Dose form
- Strength and units
- Company name
- Company roles (MAH, sponsor, manufacturer)
Authorities requiring controlled vocabulary alignment
- EMA — SPOR / IDMP
- FDA — SPL / structured data
- Swissmedic — eCTD 4.0
- Health Canada
- PMDA (Japan)
Without strong governance, organisations face
- Inconsistent submissions
- Manual reconciliation between systems
- Delays in regulatory filings
- Increasing operational risk
Three areas of regulatory data shifting simultaneously
eCTD 4.0
Stricter structured metadata expectations. Greater reliance on authority-defined controlled vocabularies. More data-driven validation outcomes that surface issues at submission time.
Explore eCTD 4.0 →IDMP & EU SPOR
Structured product and referential data becomes operational. Internal vocabularies must reconcile with external identifiers. Lifecycle traceability matters more across the product record.
Explore IDMP →Controlled Vocabularies
Multiple external "golden sets" to track across authorities. Overlapping codes with slight differences in identifiers, lifecycle definitions and usage rules create reconciliation effort across teams.
Explore CVs →Explore the regulatory data landscape
The secure portal supports deeper exploration of Health Authority vocabularies, mappings, term alignment and AI-assisted coding across multiple regulatory frameworks.